Overexpression or hyperactivation of ErbB cell-surface receptors are known to enhance the growth of various breast cancers. Though medications like Herceptin are apparently capable of halting the receptors’ signals, all patients fail to provide positive results. Scientists from the University of Pennsylvania School of Medicine claim that the protein P-Rex1 is important for signal transmission from ErbB receptors and hence can be targeted for treating cancer.
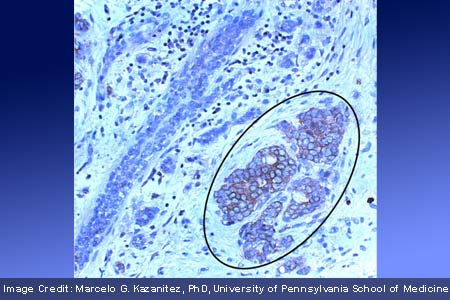
Having scrutinized breast cancer samples, it appeared that the protein is over expressed in almost 60 percent of tested samples and patients whose tumors express P-Rex1 were more likely to develop metastasis. A downstream target of the ErbB receptors seems to be vital for cancer cell proliferation, migration, and metastasis. Detailed insight into this pathway can possibly help introduce novel drugs or therapeutic approaches in the future. P-Rex1 may be present in some breast tumors, especially those that express the Her2/neu receptor or estrogen receptor and belong to the luminal subtype.
Marcelo G. Kazanietz, PhD, professor of Pharmacology at the University of Pennsylvania School of Medicine, explained, “We found that about two-thirds of the patient samples had very high levels of P-Rex1 expression in tumor cells in their lymph nodes. There seems to be a correlation between P-Rex1 expression in the tumor cells and the capacity of these cells to metastasize. And since P-Rex1 is likely to be essential for cell migration and migration is essential for metastasis, we believe blocking this pathway could reduce the risk of metastasis.â€
Researchers predict that P-Rex1 plays a pivotal role for several other cancer-promoting pathways. The receptor CXCR4 is also believed to employ P-Rex1. In spite of its utilization in numerous pathways during cancerous growth, P-Rex1 may not be expressed in many normal tissues. Therefore, targeting this protein appears beneficial for treating cancers. P-Rex1 is supposedly expressed in some subtypes of breast tumors and can serve as an excellent prototype for future personalized medicine.
The research is published in on the December 22 issue of Molecular Cell.
