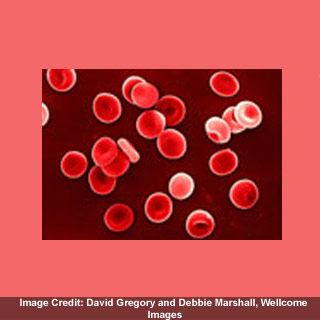
To locate genetic variants that affect blood measurements, a UK and German team, together with experts from the Wellcome Trust Sanger Institute, examined genomes of over 14 000 people. They apparently discovered 22 areas of the human genome mixed up in the development of blood cells, together with 15 that were formerly unrevealed.
They also found one genetic variant linked to platelet counts whose incidence augments the possibility of developing heart disease. This was discovered in an area of the genome already known to affect the threat of hypertension, coeliac disease and type 1 diabetes in children and young adults.
Dr Nicole Soranzo, group leader at the Wellcome Trust Sanger Institute and co-lead of the HaemGen consortium that conducted the study, commented, “We have uncovered a novel variant linking platelet counts with heart attacks. Further characterization of the regions uncovered in this study has the potential to improve our understanding how blood cell development is linked with human diseases, including blood cell cancers.â€
The experts gauged the concentration of hemoglobin, as well as the amount and quantity of red and white cells and platelets from people in UK and Germany. They then evaluated areas of the human genome caught up in blood cell development with regions linked to amplified danger of heart disease, in addition to comparing the genomes of about 10 000 people suffering from heart disease with those of 10 000 healthy people.
The genetic risk factors discovered were believed to be powerfully associated with European ancestry and the experts estimate that these became permanent in European genomes around 3400 years ago. They propose that the threat factors were passed on as they could give transporters a bigger defense against infection.
